One thing you can do is learn to read labels. You can also check the website of some very helpful non-profit associations that test botanicals to find out which products they recommend.
- How should I read labels?
- Who is looking at the quality of botanicals?
- Where can I find recommended products?
How should I read labels?
The federal government regulates the sale of botanicals with the Dietary Supplement Health and Education Act (DSHEA). This act governs what manufacturers of dietary supplements can and can't put on their labels. (See Are Botanical Medicines Safe? for more information about DSHEA.)
All labels must include:
- The term "dietary supplement" or a similar term such as "vitamin E supplement" or "herbal supplement." This lets you know the product is not subject to the laws governing drugs.
- A facts panel with information on nutrients and percent recommended daily allowance.
- A list of all other dietary ingredients in the product, including ones for which no recommended daily allowance has been established.
- The name and place of the manufacturer or distributor
Note that there are no government regulations that govern a serving size (dosage) or amount of nutrient in a dietary supplement. This is up to the manufacturer. You will want to verify the dosage with your provider, or do your own research.
Health Claims
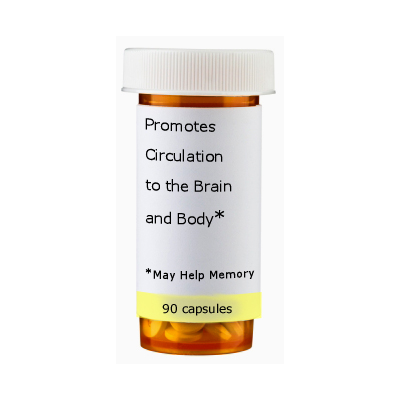 Manufacturers can make statements about how the herb affects structure and function, but they cannot make health claims that the product cures, prevents, or mitigates disease. For example, a manufacturer could say "promotes healthy circulation in the brain; may help memory," but could not say, "prevents senile dementia."
Manufacturers can make statements about how the herb affects structure and function, but they cannot make health claims that the product cures, prevents, or mitigates disease. For example, a manufacturer could say "promotes healthy circulation in the brain; may help memory," but could not say, "prevents senile dementia."
When claims are included on product labels, a disclaimer must be included. Typical wording might be: "These statements have not been evaluated by the Federal Drug Administration. This product is not intended to diagnose, cure, treat, or prevent any disease."
This wording is required because the FDA does not evaluate dietary supplements and approve them prior to market release, as they do with drugs. If a label did claim that the product was a treatment or prevention for a specific disease or condition, it would be considered an unapproved and thus illegal drug.
These labeling requirements are confusing to many people because they are using dietary supplements to treat or prevent disease (and there is much research to support them doing so in many cases). Because the labels aren't helpful, you have to do your own research to determine how to effectively, and appropriately, use each botanical.
Non-Government Items
Several organizations offer quality evaluation services to manufacturers. Products that pass these evaluations can include a certification logo on the label. See Where Can I Find Recommended Products? below.
Who is looking at the quality of botanicals?
The quick answer is the FDA and various non-profit associations. Both are working on the quality of botanicals (dietary supplements) by looking at the manufacturing standards used to produce the products. These are referred to as good manufacturing process (GMP) standards.
Currently, the botanical products industry is legally bound to the same federally mandated good manufacturing process standards as the food industry. These GMPs assure that controlled, sanitary manufacturing practices are in place and that the resulting products contain what is on the label.
Recently approved GMP standards for dietary supplements
The original DSHEA legislation, passed in 1994, included provisions for the development of standards for Good Manufacturing Practice of dietary supplements. These are similar, but not identical, to the current FDA standards for foods. It took 13 years of discussions between the FDA and industry trade associations, such as the American Herbal Products Association, United States Pharmacopeia, and NSF International, to develop Good Manufacturing Practice guidelines that are specific to dietary supplements. These guidelines focus on practices that ensure that dietary supplements are pure and contain the ingredients listed, in the strength indicated.
This process has now been completed, and in June 2007 the FDA issued its final rule on the implementation of Good Manufacturing Practice standards for dietary supplements. As of June 2010, all manufacturers of dietary supplements must be in compliance with these standards.
Where can I find recommended products?
The organizations listed below certify that the products they recommend:
- Actually contain the ingredients listed on the label in the declared amount and strength
- Do not exceed contamination limits
- Comply with good manufacturing practices
In general, these associations review and evaluate the manufacturer's quality control, manufacturing data, and laboratory samples. They also conduct audits of good manufacturing practices.
United States Pharmacopeia
USP is an independent, not-for-profit organization. Manufacturers can voluntarily submit their products to USP for evaluation. Those that pass can display the USP logo.
See www.usp.org for more information on the program, a list of manufacturers (and brands) that participate in the USP evaluation program, and a list of stores where you can buy the certified products.
NSF International
NSF International is an independent, not-for-profit organization, committed to making the world a safer place for consumers. Products that pass the NSF program can carry the NSF certification logo.
For more information, see www.nsf.org. The site allows you to search a particular brand of botanicals to see if it is in the NSF database of certified dietary supplements.
ConsumerLabs.com
ConsumerLabs.com is a certification company that tests products voluntarily submitted by dietary supplement manufacturers. In the past five years, CL has tested more than 1,200 products, representing more than 250 different brands.
Note that you have to be a paid subscriber to get some of their information and the site accepts ads from manufacturers. But there is a lot of valuable information, including good information on recalls.
For more information, see http://www.consumerlabs.com/.
Another Resource
Another useful resource is the website of the Dietary Supplement Quality Initiative.
Barrett, M. (Ed.) (2004). The Handbook of Clinically Tested Herbal Remedies, Vol. 1 and 2. New York: Haworth Press, Inc.
Cavalier, C., Blumenthal, M. (2007). Review of FDA's final GMPs for dietary supplements. Herbalgram, 76, 58-61.
Center for Food Safety and Applied Nutrition Dietary Supplement Overview: http://www.cfsan.fda.gov/~dms/supplmnt.html
Dietary Supplement Health and Education Act: http://www.health.gov/dietsupp/ch1.htm
United States Pharmacopeia: www.usp.org
NSF International: www.nsf.org
ConsumerLabs: http://www.consumerlabs.com/
Dietary Supplement Quality Initiative: http://www.supplementquality.com/links/monographs.html


